In the intricate workings of nature, photosynthesis serves as a fundamental process that allows plants and some bacteria to convert light energy into chemical energy. This natural phenomenon has inspired technological advancements, most notably in the realm of solar energy, where photovoltaics convert solar radiation into electricity. At the heart of both processes lies the movement of electrons and the transfer of charges at the molecular level.
Understanding the molecular dynamics behind these reactions is crucial, particularly how molecules redistribute electronic density upon absorbing light. This ultrafast reconfiguration occurs in a matter of femtoseconds and attoseconds, and involves complex quantum effects. The capability to observe these minute and rapid electronic processes not only sheds light on intrinsic physical mechanisms, but also opens avenues for engineering materials with enhanced properties for energy applications.
Researchers have recently harnessed the power of ultrashort ultraviolet pulses from high-order harmonic generation or free electron laser facilities to delve deeper into the phenomena of photoionization and electron transfer. These advanced techniques enable observation of molecular responses on exceedingly brief timescales, ranging from 10^-15 seconds (femto) to 10^-18 seconds (atto). However, despite the progress achieved, many questions remain unanswered regarding the preliminary stages of electron and charge transfer following prompt photoionization.
To address this research gap, a pioneering study was conducted by a collaborative team from various institutions, including the Politecnico di Milano and Madrid Institute for Advanced Studies in Nanoscience (IMDEA Nanociencia). Their findings, published in Nature Chemistry, mark a significant advancement in our understanding of charge transfer dynamics, specifically within donor-acceptor molecular systems.
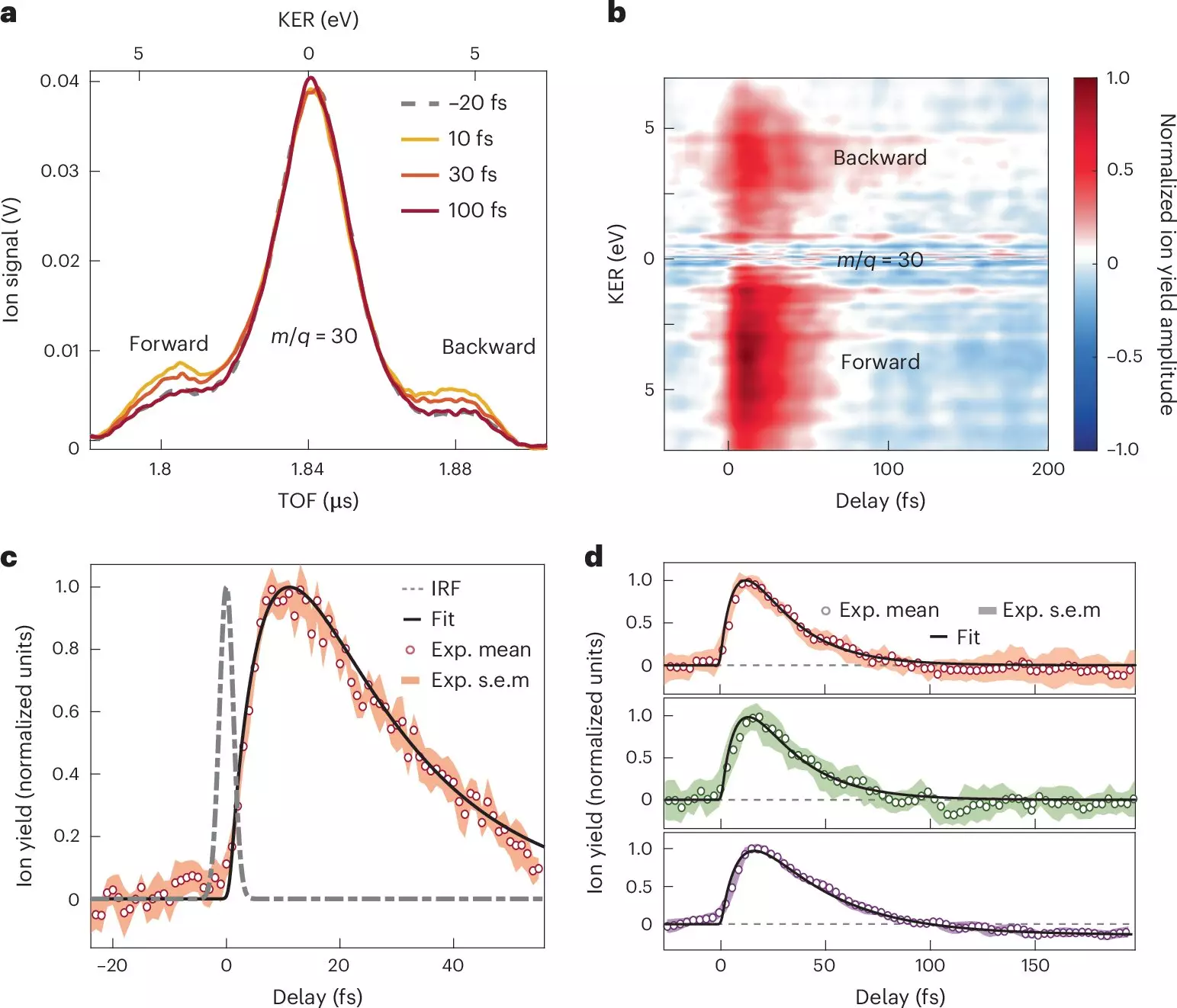
Using attosecond extreme-ultraviolet pulses, the researchers were able to investigate the ultrafast dynamics of nitroaniline molecules with an unprecedented level of precision. By employing an innovative combination of attosecond extreme-ultraviolet-pump and few-femtoseconds infrared-probe spectroscopy, along with sophisticated many-body quantum chemistry calculations, the team unveiled the rapid processes underpinning charge transfer.
The key findings indicated that electron transfer occurs incredibly swiftly—from the electron donor amino group—within less than 10 femtoseconds. This rapid transfer is coupled with a coordinated motion between nuclei and electrons, laying the groundwork for a relaxation phase that unfolds over a sub-30-femtosecond timescale. The interaction between the energetic electron and the molecular structure plays a critical role in this process.
The study emphasizes the significant influence of electron-nuclear coupling on the dynamics of donor-acceptor molecules, particularly in response to the excitation caused by photoionization. The researchers meticulously tracked the time it takes for charge to migrate from the donor unit to the adjacent bond linking it to a benzene ring. Furthermore, the structural transformations necessary for this charge migration were rigorously mapped, yielding critical insights into the dynamics of molecular interactions.
The implications of these findings extend beyond mere experimental observations. The authors suggest that their research could enrich the existing theoretical frameworks and methodologies used to qualitatively characterize charge movement in organic compounds. This study not only enhances our understanding of chemical interactions at a fundamental level but can potentially inform the development of materials with optimized electronic properties.
As we look forward, the revelations made by this research pave the way for further exploration in the field of attosecond science. By elucidating the principles of molecular dynamics, researchers can explore targeted modifications in chemical structures that could lead to improved functionality in areas such as organic photovoltaics and molecular electronics.
Overall, as scientists continue to unravel the complexities of molecular interactions through ultrafast techniques, we stand on the cusp of transforming our theoretical and practical grasp of electron dynamics, which could lead to revolutionary advancements in energy conversion technologies and beyond.